ENPP1 and pyrophosphate: the biology behind the platform
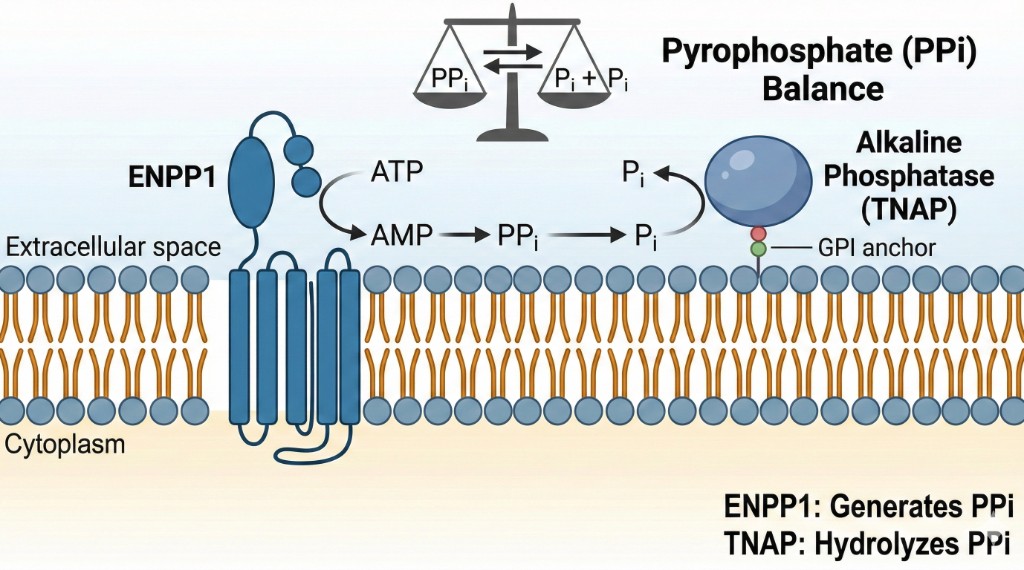
ENPP1 generates PPi from ATP; TNAP hydrolyzes PPi. Their balance governs mineralization.
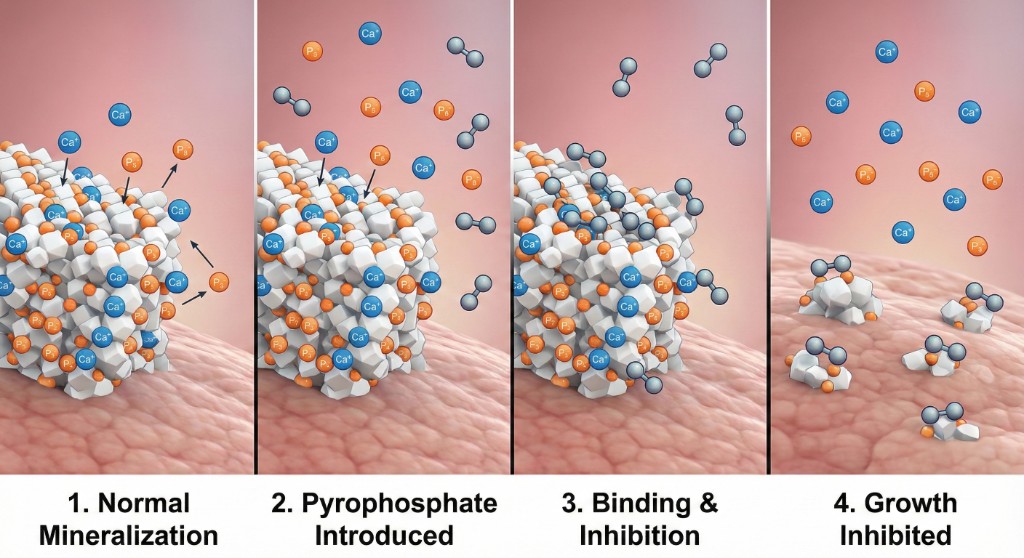
Pyrophosphate binds to mineralized surfaces, inhibiting further crystal growth.
ENPP1 is the only enzyme that generates extracellular pyrophosphate (PPi), a potent endogenous inhibitor of hydroxyapatite mineralization. Research from the Braddock laboratory at Yale demonstrated that ENPP1 regulates bone mass through both catalysis-dependent and catalysis-independent signaling pathways (Zimmerman et al., J Bone Miner Res 2022). Because pyrophosphate balance governs how and where mineralization occurs, ENPP1 biology has relevance across multiple disorders involving abnormal calcification, impaired tissue repair, or crystal deposition.
Foundational research established that pyrophosphate is the central regulator of acellular cementum formation, with cementoblasts actively modulating local PPi concentrations to control mineralization on tooth root surfaces (Foster et al., PLoS ONE 2012). Subsequent studies showed that counter-regulatory phosphatases TNAP and NPP1 (ENPP1) temporally regulate cementogenesis, with NPP1 expression increasing as cementum matures to restrict further apposition (Foster et al., Int J Oral Sci 2015). Work on hypophosphatasia demonstrated that correcting the phosphate/pyrophosphate ratio rescues mineralization in periodontal ligament cells (Rodrigues et al., J Periodontol 2011) and that HPP-associated pyrophosphate imbalance disrupts odontoblast gene expression and dentin mineralization (Rodrigues et al., J Endod 2012). Together, these findings shaped the thesis that targeted modulation of ENPP1 biology may support tissue restoration across multiple clinical contexts.
A shared pathway across distinct clinical conditions
Petragen is focused on diseases in which pathological mineralization contributes to progression or unmet clinical burden. This includes periodontal disease, affecting nearly 36 million people in the U.S. where current treatments offer minimal benefit; hypophosphatasia, where current biological treatments can cost up to $2 million per year and require near daily injections; and CPPD, a crystal arthropathy affecting nearly 10 million people with limited disease-modifying options.
Mechanism first. Development minded.
Our scientific strategy combines target biology, translational disease selection, formulation and delivery thinking, and practical development paths. We prioritize programs where mechanism, clinical need, and product strategy align.
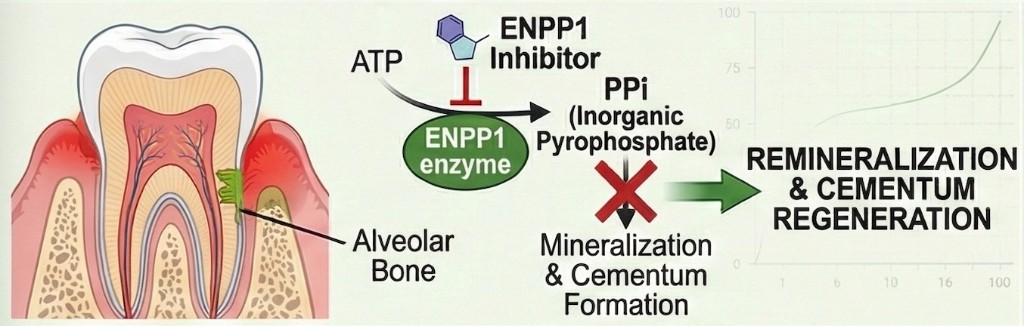
Periodontal disease
ENPP1 generates pyrophosphate that regulates cementum formation on tooth root surfaces. In periodontal disease, loss of cementum and alveolar bone leads to tooth loosening and loss. Petragen is developing a locally delivered therapeutic that modulates ENPP1 biology to support cementum regeneration and restore the tooth-bone attachment apparatus, targeting the direct pathophysiology rather than downstream inflammation.
Explore Periodontal disease →
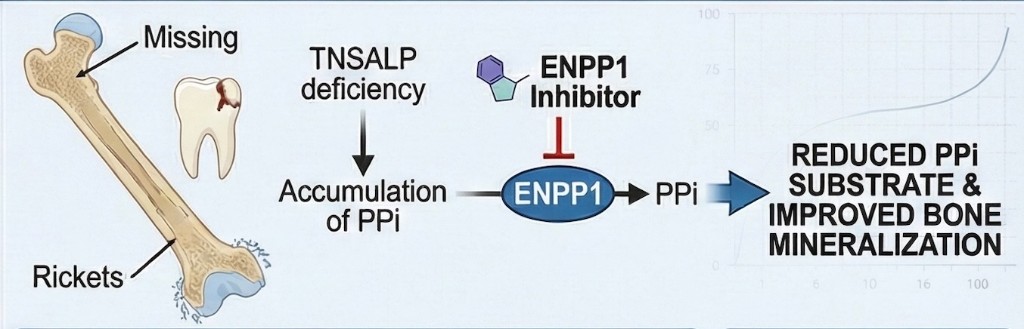
Hypophosphatasia (HPP)
HPP is caused by mutations in the ALPL gene encoding alkaline phosphatase (TNAP), the counter-regulatory enzyme to ENPP1 that hydrolyzes pyrophosphate. When TNAP is deficient, excess pyrophosphate accumulates and inhibits skeletal and dental mineralization. Petragen is developing a small molecule approach that targets this phosphate/pyrophosphate imbalance to restore normal mineralization without the burden and cost of current biological therapies.
Explore HPP →
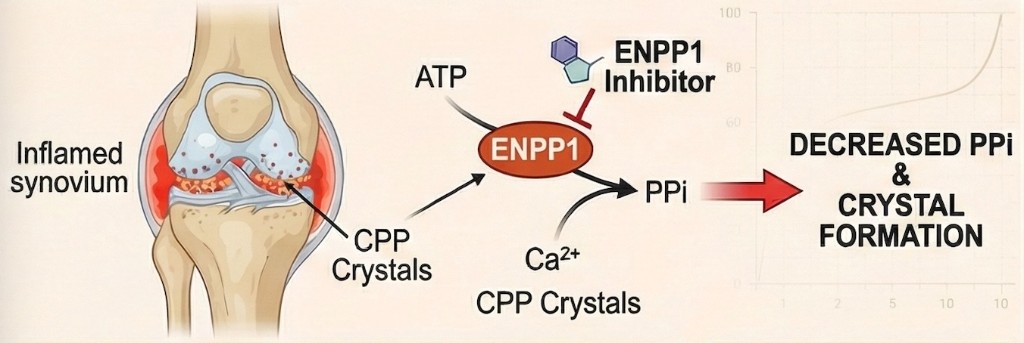
Calcium pyrophosphate disease (CPPD)
CPPD arises from the deposition of calcium pyrophosphate crystals in cartilage and periarticular tissues, directly driven by excess extracellular pyrophosphate. ENPP1 is the primary enzymatic source of this pyrophosphate. Petragen is targeting the ENPP1-mediated overproduction of pyrophosphate to reduce crystal formation at its source, aiming to deliver a genuinely disease-modifying therapy rather than treating downstream inflammation alone.
Explore CPPD →
Publications & presentations
-
Catalysis-independent ENPP1 protein signaling regulates mammalian bone mass
Zimmerman K, Li X, von Kroge S, Stabach P, Lester ER, Chu EY, Srivastava S, Somerman MJ, Tommasini SM, Busse B, Schinke T, Carpenter TO, Oheim R, Braddock DT
Journal of Bone and Mineral Research · 2022
Demonstrates that ENPP1 regulates bone mass through catalysis-independent protein signaling pathways, uncovering a dual role for ENPP1 beyond its enzymatic function and providing the mechanistic basis for Petragen's platform.
-
Counter-regulatory phosphatases TNAP and NPP1 temporally regulate tooth root cementogenesis
Foster BL, Ao M, Willoughby C, Soenjaya Y, Holm E, Lukashova L, Tran AB, Wimer HF, Zerfas PM, Nociti FH Jr, Kantovitz KR, Quan BD, Sone ED, Goldberg HA, Somerman MJ
International Journal of Oral Science · 2015
Defines how TNAP and NPP1 act as counter-regulatory phosphatases to control cementum formation on tooth roots, directly informing Petragen's approach to periodontal regeneration.
-
Hypophosphatasia-associated deficiencies in mineralization and gene expression in cultured dental pulp cells obtained from human teeth
Rodrigues TL, Foster BL, Silverio KG, Martins L, Casati MZ, Sallum EA, Nociti FH Jr, Somerman MJ
Journal of Endodontics · 2012
Demonstrates that hypophosphatasia disrupts mineralization and odontoblast gene expression in human dental pulp cells, connecting ENPP1/pyrophosphate biology to the HPP dental phenotype.
-
Central role of pyrophosphate in acellular cementum formation
Foster BL, Nagatomo KJ, Tso HW, Tran AB, Nociti FH Jr, Narisawa S, Yadav MC, McKee MD, Millan JI, Somerman MJ
PLoS ONE · 2012
Establishes pyrophosphate as the central regulator of acellular cementum formation, showing that cementoblasts actively tune local PPi levels to control mineralization on tooth root surfaces.
-
Correction of hypophosphatasia-associated mineralization deficiencies in vitro by phosphate/pyrophosphate modulation in periodontal ligament cells
Rodrigues TL, Nagatomo KJ, Foster BL, Nociti FH Jr, Somerman MJ
Journal of Periodontology · 2011
Shows that correcting the phosphate/pyrophosphate ratio rescues mineralization in HPP periodontal ligament cells, supporting the therapeutic rationale for modulating ENPP1 activity in periodontal disease.
-
Hypercementosis associated with ENPP1 mutations and GACI
Thumbigere-Math V, Alqadi A, Guzman Vega F, Owczarczak-Garstecka SC, Tso HW, Chu EY, Chojnowski J, Foster BL, Somerman MJ, Braddock DT
Journal of Dental Research · 2018
Supports the biological connection between ENPP1 dysfunction and cementum accumulation in human GACI patients, helping frame the rationale for periodontal regeneration.
-
Genetic and pharmacologic modulation of cementogenesis via pyrophosphate regulators
Chu EY, Chavez MB, Hu Y, Foster BL, Somerman MJ, Braddock DT
Bone · 2020
Provides direct evidence that genetic and pharmacologic modulation of pyrophosphate regulators influences cementogenesis, validating the target mechanism for Petragen's periodontal program.
Important disclosure
This website may include forward-looking statements regarding investigational programs that are subject to risks and uncertainties.