Disease overview
Periodontal Disease
One of the most common chronic diseases, affecting nearly 36 million people in the U.S.
Overview
Periodontal diseases encompass a range of inflammatory conditions affecting the tissues that support the teeth, including the gingiva, periodontal ligament, cementum, and alveolar bone. Disease typically begins as gingivitis and, if untreated, can progress to chronic periodontitis, characterized by irreversible loss of connective tissue attachment and alveolar bone. Advanced chronic periodontitis may affect 10-15% of the global population. The disease is often painless and can reach advanced severity before detection. Hallmarks include periodontal pocket formation, clinical attachment loss, and radiographic bone loss. Diagnosis relies on clinical parameters including probing depth, bleeding on probing, and radiographic findings. Disease is initiated by dysbiosis of the microbial biofilm on teeth, with the host inflammatory response determining the course of tissue destruction.
Periodontal disease leads to loss of the cementum, periodontal ligament, and alveolar bone that anchor teeth. ENPP1 modulation targets the mineralization biology underlying attachment loss.
Clinical burden
Periodontal disease causes progressive destruction of the tooth-supporting apparatus, leading to tooth mobility, tooth loss, impaired mastication, and significant quality-of-life impact. Comorbidities including diabetes mellitus and cardiovascular disease can worsen the inflammatory state. Current treatment relies on scaling and root planing, surgical intervention, and long-term maintenance, but existing therapies offer minimal disease-modifying benefit, with fractions of a millimeter improvement in key clinical endpoints like pocket depth.
Current care gap
Despite being one of the most prevalent chronic diseases, there are virtually no disease-modifying therapeutics for periodontitis. Current management centers on mechanical debridement, adjunctive antibiotics, and surgical procedures to reduce pocket depth and promote regeneration. Long-term success depends heavily on patient adherence to maintenance therapy. Novel approaches under investigation include protein-based, gene-based, and cell-based regenerative strategies, but none has achieved widespread clinical adoption. The fundamental gap remains the absence of a therapeutic that can restore the cementum and periodontal ligament attachment that is lost to disease.
Role of ENPP1 in periodontal disease
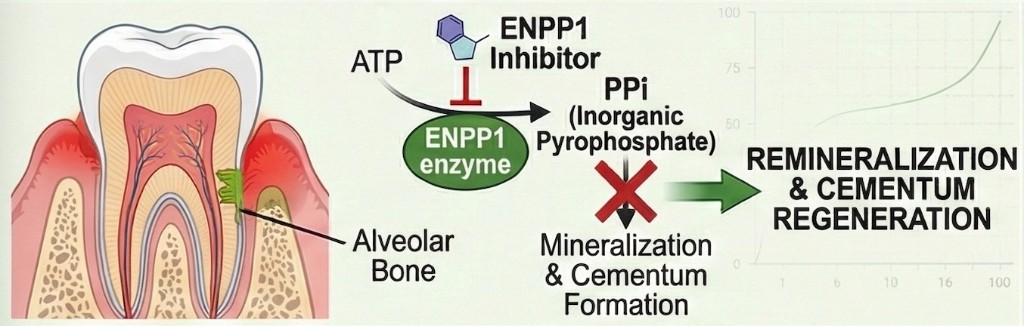
ENPP1 generates the pyrophosphate that regulates cementum formation on tooth root surfaces. Foundational research demonstrated that pyrophosphate is the central regulator of acellular cementum formation, with cementoblasts actively modulating local PPi concentrations to control mineralization (Foster et al., PLoS ONE 2012). Counter-regulatory phosphatases TNAP and NPP1 (ENPP1) temporally regulate cementogenesis, with NPP1 expression increasing as cementum matures to restrict further apposition (Foster et al., Int J Oral Sci 2015). In periodontal disease, loss of cementum and alveolar bone compromises the tooth-bone attachment apparatus. Petragen is developing a locally delivered therapeutic that modulates ENPP1 biology to support cementum regeneration and restore periodontal attachment, targeting the direct pathophysiology rather than downstream inflammation alone.
Petragen perspective
Petragen believes it can be best in class for this large and underserved indication by targeting the mechanism at the point where disease actually occurs. Our locally delivered approach is grounded in ENPP1 biology and the biological rationale for restoring the cementum and periodontal ligament structures that anchor teeth to bone. This approach is aligned with the periodontal treatment workflow and designed for practical clinical adoption.
References
- Kinane DF, Stathopoulou PG, Papapanou PN. Periodontal diseases. Nature Reviews Disease Primers. 2017;3:17038.
- Foster BL et al. Central role of pyrophosphate in acellular cementum formation. PLoS ONE. 2012;7(6):e38393.
- Foster BL et al. Counter-regulatory phosphatases TNAP and NPP1 temporally regulate tooth root cementogenesis. Int J Oral Sci. 2015;7(1):27-41.
