Disease overview
Calcium Pyrophosphate Disease (CPPD)
Crystal deposition disease affecting nearly 10 million people in the U.S.
Overview
CPPD is a common but underdiagnosed crystal arthropathy caused by the deposition of calcium pyrophosphate dihydrate (CPP) crystals in articular cartilage and periarticular tissues. The disease is associated with excessive local production of extracellular inorganic pyrophosphate (PPi). CPPD shows equal gender distribution and increases markedly with age, with evidence of articular chondrocalcinosis in almost half of individuals aged 80-89 years and prevalence doubling with each decade after age 60. The most recognized manifestation is acute CPP crystal arthritis, presenting as acute attacks of synovitis that clinically resemble gout flares, but chronic CPPD-associated arthropathies including those mimicking osteoarthritis or rheumatoid arthritis also exist. Risk factors include age, previous joint trauma, osteoarthritis, and metabolic conditions including hypophosphatasia and hyperparathyroidism. Diagnosis is best made by identification of positively birefringent crystals in synovial fluid.
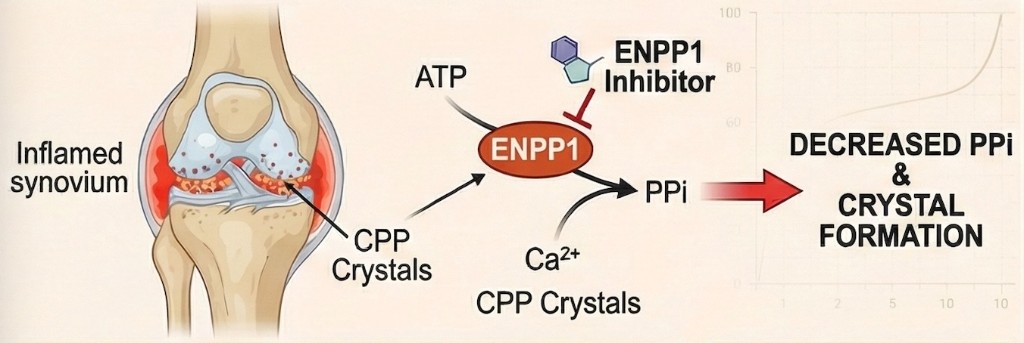
In CPPD, ENPP1 inhibition may decrease pyrophosphate levels and reduce calcium pyrophosphate crystal formation in affected joints.
Clinical burden
CPPD causes recurrent acute flares, chronic joint pain, reduced mobility, and progressive structural joint damage. The similarity to gout frequently leads to misdiagnosis. Chronic forms can cause cartilage degradation, osteophytosis, and subchondral bone changes similar to osteoarthritis. CPP and BCP crystals activate the NLRP3 inflammasome, driving IL-1 production, neutrophil recruitment, and tissue injury. No drug exists to prevent crystal deposition, permit crystal dissolution, or specifically target the pathogenic effects of CPP crystals.
Current care gap
Current management is limited to symptomatic treatment with NSAIDs, colchicine, and corticosteroids for acute flares, with few options for chronic disease. In contrast to gout, where urate-lowering therapies can dissolve crystals, no drug exists that can dissolve CPP crystals or prevent their formation. Hydroxychloroquine and methotrexate have shown limited benefit in uncontrolled settings. IL-1 inhibitors provide some relief but do not address the underlying crystal deposition. The fundamental unmet need is a therapy that reduces pyrophosphate-driven crystal formation at its source.
Role of ENPP1 in CPPD
ENPP1 is the primary enzymatic source of the extracellular pyrophosphate that, when present in excess, combines with calcium to form the CPP crystals that drive CPPD pathology. Overexpression of ANKH (which also regulates extracellular PPi transport) in osteoarthritic chondrocytes results in excess extracellular PPi generation favouring CPP crystal formation. Gain-of-function mutations in ANKH are associated with familial CPPD disease, directly linking pyrophosphate dysregulation to crystal deposition. Drugs targeting the pyrophosphate pathway may limit crystal deposition in CPP-related arthropathies. Petragen is targeting the ENPP1-mediated overproduction of pyrophosphate to reduce crystal formation at its enzymatic source.
Petragen perspective
Petragen sees CPPD as an opportunity to deliver a genuinely disease-modifying therapy by targeting the mechanism at the point where crystal deposition occurs. Rather than treating downstream inflammation alone, our approach aims to reduce the excess pyrophosphate that drives CPP crystal formation, staying within a modality that patients and physicians are accustomed to.
References
- McCarthy GM, Dunne A. Calcium crystal deposition diseases - beyond gout. Nature Reviews Rheumatology. 2018;14:592-602.
- Rosenthal AK, Ryan LM. Calcium pyrophosphate deposition disease. N Engl J Med. 2016;374:2575-2584.
- Collins MT et al. Skeletal and extraskeletal disorders of biomineralization. Nature Reviews Endocrinology. 2022;18:473-489.
